# Week 2 workshop - Tidying data
# Written by Nicholas Wu, 06/11/2024, Murdoch University
# Load packages
library(tidyverse)
library(visdat)
# Set your working directory
setwd("YOUR-DIRECTORY")
# Load your data
plant_task_clean_data <- read_csv("data/plant_task_clean_data.csv")2 Tidy Data
Week 2 - Tidying data and clean code
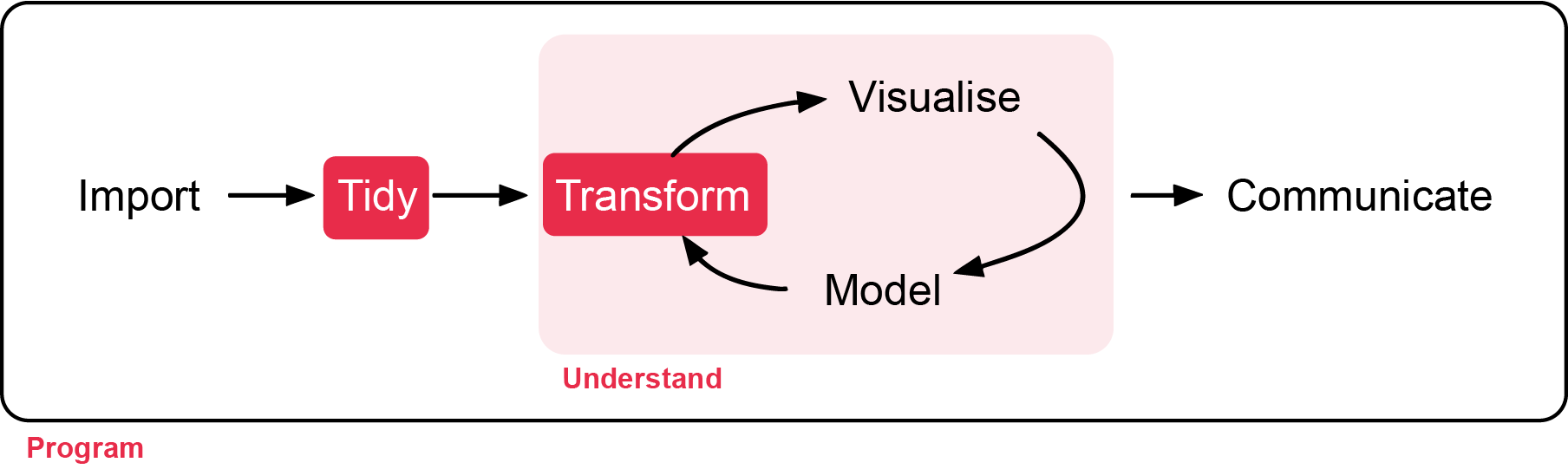
In this workshop, you will learn how transform raw data to something usable for analysis, and get familiar with clean code. Workshop materials are available in the github repository ECS200.
Background reading
Sometimes, it is rare that you get the data in exactly the right form you need from the raw data you import into R. Often you’ll need to create some new variables or summaries, or maybe you just want to rename the variables or reorder the observations in order to make the data a little easier to work with. In this workshop you are going to learn the five key dplyr functions (part of tidyverse) that allow you to solve the vast majority of your data manipulation challenges:
- Pick observations by their values
filter(). e.g. keeping rows with values higher than 100. - Reorder the rows
arrange(). e.g. rearrange a numeric column by the largest values. - Pick variables by their names
select(). e.g. keeping selected columns only. - Create new variables with functions of existing variables
mutate(). e.g. the sum of values from two numeric columns. - Collapse many values down to a single summary
summarise(). e.g. getting the overall mean of the dataset.
These can all be used in conjunction with group_by() which changes the scope of each function from operating on the entire dataset to operating on it group-by-group. These six functions provide the verbs for a language of data manipulation.
All verbs work similarly:
- The first argument is a data frame.
- The subsequent arguments describe what to do with the data frame, using the variable names (without quotes).
- The result is a new data frame.
Together these properties make it easy to chain together multiple simple steps to achieve a complex result. Let’s dive in and see how these verbs work.
Study context
In order to know whether our imported data requires transformation before analysis, we need to know what we are analysing. Let’s stick with a familiar dataset, the plant_calcium.csv from this study.
The plant_calcium.csv dataset contains observations on sugar maple seedlings in untreated and calcium-treated watersheds at Hubbard Brook Experimental Forest in New Hampshire, USA.
Growth of sugar maples (Acer saccharum), known for their maple syrup and iconic leaf shape, can be stunted due to soil acidification from prolonged acid rain, which leaches calcium - a nutrient important for plant growth - from soils and stresses maple seedlings.
To investigate the impact of soil calcium supplementation on sugar maple seedling growth, researchers at the Hubbard Brook Long Term Ecological Research (LTER) site recorded “general sugar maple germinant health by height, leaf area, biomass, and chlorophyll content” (Juice et al., 2006) for seedlings in untreated and previously calcium-treated watersheds (Peters et al., 2004). By comparing seedling growth in calcium-treated (W1) versus untreated (Reference) watersheds, calcium impacts on sugar maple seedling growth can be explored.

Data Manipulation
Before we start, load your previous assessment workflow with the plant_task_data_clean.csv dataset (errors removed). Here, we will call the imported as plant_task_clean_data.
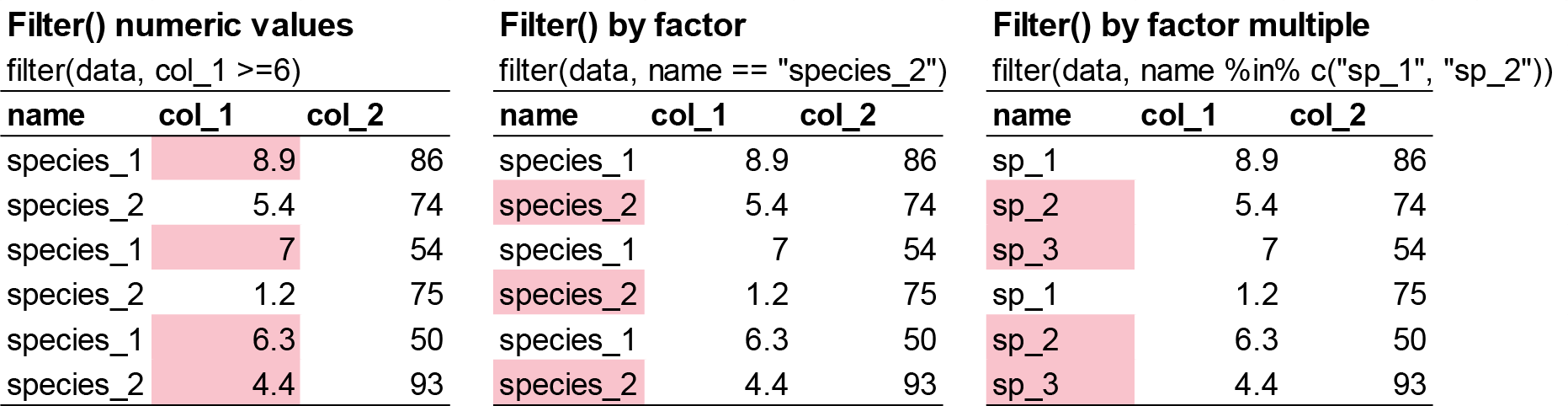
filter() allows you to subset observations based on their values. The first argument is the name of the data frame. The second and subsequent arguments are the expressions that filter the data frame. For example, we can select all values for the year 2004 for the calcium-treated sites (W1):
# filter function by two columns. A numeric and a categorical.
# Otion 1
filter(plant_task_clean_data, year == 2004, watershed == "W1")
# Option 2 using pipes %>%
plant_task_clean_data %>%
filter(year == 2004, watershed == "W1")You can also filter rows by more one variable in a column using x %in% y logic. This will select every row where x is one of the values in y. For example, we can select all values in transect R1 and W1-1 only:
# filter function for the transect column by "R1" and "W1-1"
# Option 1
filter(plant_task_clean_data, transect %in% c("R1", "W1-1"))
# Option 2 using pipes %>%
plant_task_clean_data %>%
filter(transect %in% c("R1", "W1-1"))Lastly, you can filter by number thresholds. e.g. filtering out extreme values (i.e. for data quality checks), or filter within a range of values. For example, we can select all stem_dry_mass values below 0.9 or between 0.01-0.02:
# filter function by stem_dry_mass values below 0.9
filter(plant_task_clean_data, stem_dry_mass <= 0.9)
# Option 2 using pipes %>%
plant_task_clean_data %>%
filter(stem_dry_mass <= 0.9)
# filter function by stem_dry_mass values between 0.01 and 0.02
filter(plant_task_clean_data, stem_dry_mass > 0.01 & stem_dry_mass < 0.02)
# Option 2 using pipes %>%
plant_task_clean_data %>%
filter(stem_dry_mass > 0.01 & stem_dry_mass < 0.02)Other possible functions and operators within the filter() function can be found here.
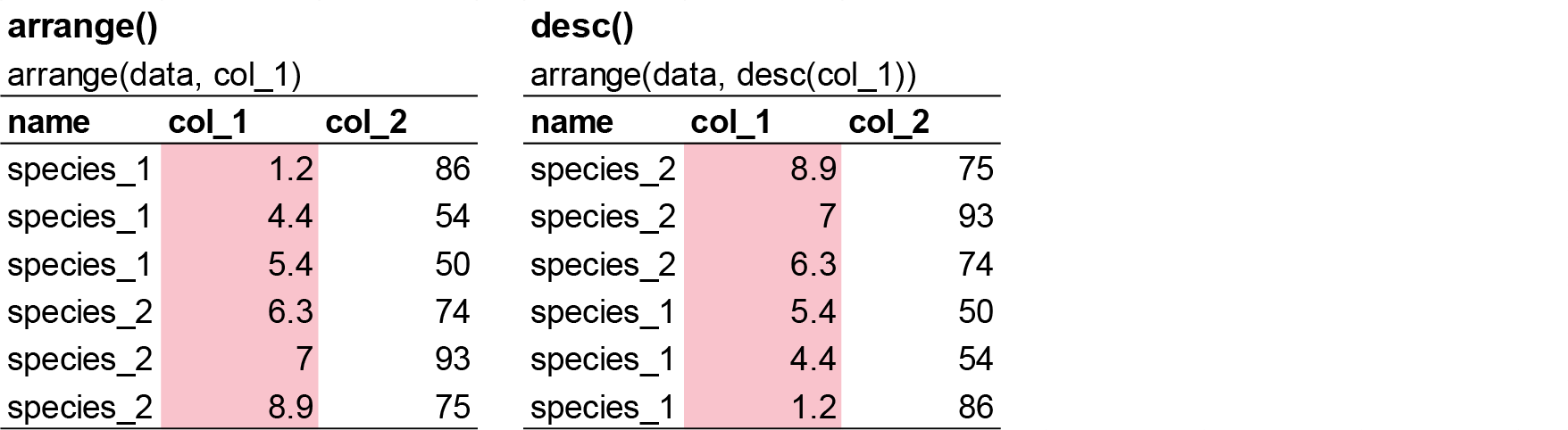
arrange() works similarly to filter() except that instead of selecting rows, it changes their order. It takes a data frame and a set of column names (or more complicated expressions) to order by. If you provide more than one column name, each additional column will be used to break ties in the values of preceding columns:
# Order the dataset by the ascending stem_length value (lowest in top row to highest in bottom row)
arrange(plant_task_clean_data, stem_length)
# Option 2 using pipes %>%
plant_task_clean_data %>%
arrange(stem_length)
# Order the dataset by the descending stem_length value (highest in top row to lowest in bottom row)
arrange(plant_task_clean_data, desc(stem_length))
# Option 2 using pipes %>%
plant_task_clean_data %>%
arrange(desc(stem_length))Note: Missing values are always sorted at the end.
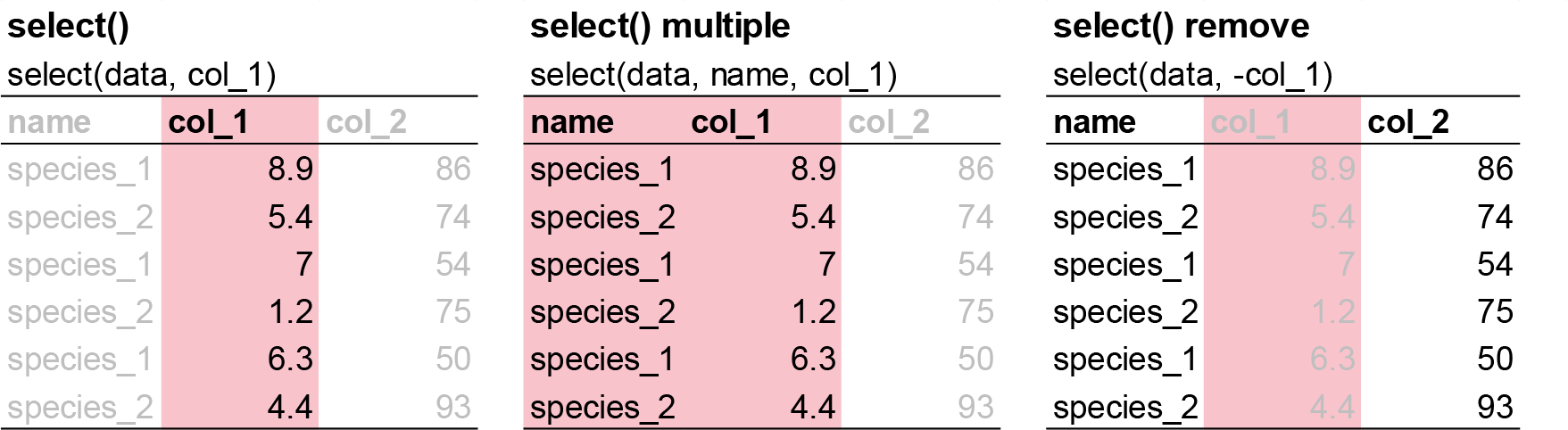
It’s not uncommon to get datasets with hundreds or even thousands of variables. In this case, the first challenge is often narrowing in on the variables you’re actually interested in. select() allows you to rapidly zoom in on a useful subset using operations based on the names of the variables.
# Select only three varaibles to keep.
select(plant_task_clean_data, year, watershed, stem_dry_mass)
# Option 2 using pipes %>%
plant_task_clean_data %>%
select(year, watershed, stem_dry_mass)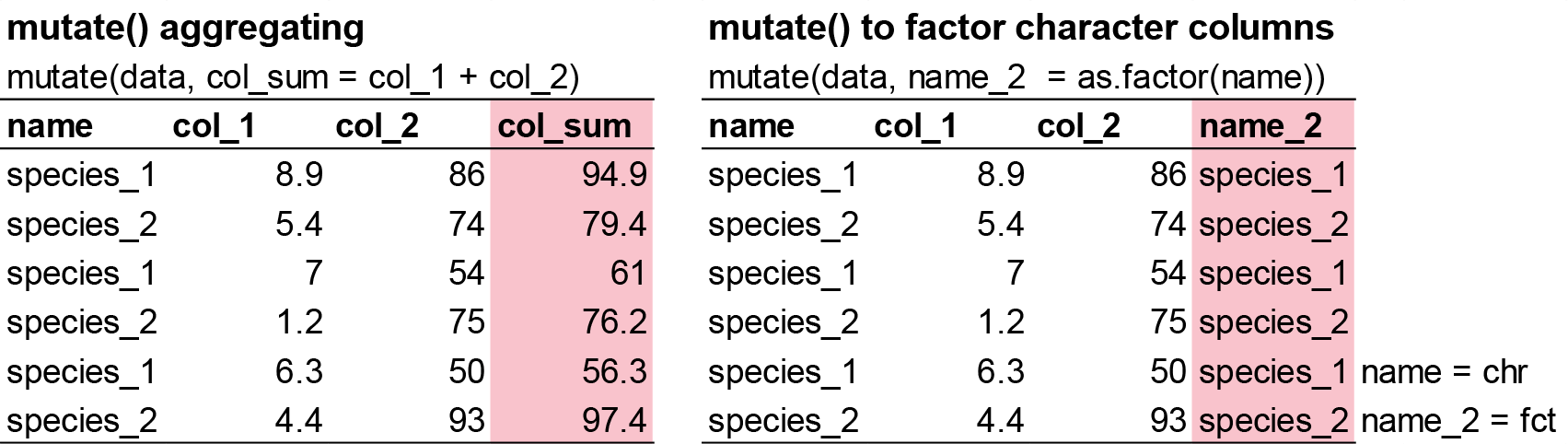
Besides selecting sets of existing columns, it’s often useful to add new columns that are functions of existing columns. That’s the job of mutate().
mutate() always adds new columns at the end of your dataset so we’ll start by creating a narrower dataset so we can see the new variables.
# Calculate the total dry mass from leaf_dry_mass and stem_dry_mass and correct the unit from kilogram to gram
plant_task_clean_data <- mutate(plant_task_clean_data,
total_dry_mass = leaf_dry_mass + stem_dry_mass,
total_dry_mass_g = total_dry_mass * 1000) # convert kilogram to gram
# Option 2 using pipes %>%
plant_task_clean_data <- plant_task_clean_data %>%
mutate(total_dry_mass = leaf_dry_mass + stem_dry_mass,
total_dry_mass_g = total_dry_mass * 1000)
The last key verb is summarise(). It collapses a data frame to a single row. However, summarise() is not terribly useful unless we pair it with group_by(). This changes the unit of analysis from the complete dataset to individual groups. Then, when you use the dplyr verbs on a grouped data frame they’ll be automatically applied “by group”. For example, if we applied exactly the same code to a data frame (plant_task_clean_data) grouped by watershed, we get the average values per treatment:
# Get the mean total_dry_mass_g by treatment groups using pipes
plant_task_clean_data %>%
group_by(watershed) %>%
summarise(mean = mean(total_dry_mass_g))What happens if we try to summarise a dataset with NA’s inside?
# Here is an example dataset
NA_data <- data.frame(site = c("A", "B", "A", "B", "A", "B"),
species = c(3, 4, 6, 2, 4, NA))
# Get the mean species by site
NA_data %>%
group_by(site) %>%
summarise(mean = mean(species))
# Try this now with na.rm = TRUE
NA_data %>%
group_by(site) %>%
summarise(mean = mean(species, na.rm = TRUE))That’s because aggregation functions obey the usual rule of missing values: if there’s any missing value in the input, the output will be a missing value. Fortunately, all aggregation functions have an na.rm argument which removes the missing values prior to computation. If your dataset has NAs, the na.rm = TRUE argument will be helpful.
Exercise (15 min)
🧪 Use the previous functions to complete the following task for the fake_data below
fake_data <- data.frame(
site = c("A", "B", "C", "A", "B", "A", "B", "C", "A", "B", "A", "B", "A", "B", "C", "C", "B", "A", "b", "C", "B", "C", "C", "A", "A", "A", "A", "B", "C", "C", "C"),
species_1 = as.integer(c(7, 25, 30, 50, 38, 13, 5, 31, 34, 22, 41, 25, 22, 37, 39, 13, 45, 10, 40, 7, 28, 36, 19, 2, 12, 5, 16, 38, 27, 44, 44)),
species_2 = as.integer(c(9, 8, 1 , 7, 9 , 9, 4, 8, 1, 7, 1, 6, 4, 4, 7, 2, 8, 7, 2, 9, 9, 2, 6, 8, 6, 6, 17, 1, 8, 4, 4)),
species_3 = as.integer(c(190, 160, 70, 430, 310, 99, 530, 420, 371, 357, 198, 171, 463, 124, 254, 484, 435, 409, 122, 305, 410, 162, 473, 200, 401, 273, 421, 419, 293, 487, 487)),
species_4 = as.integer(c(2, 0, 4, 8, 6, 3, 7, 4, 2, NA, 35, 10, 6, 4, 8, 1, 1, 0, NA, 4, 6, 8, 10, 6, 2, 5, 10, 7, NA, NA, NA))
)- Conduct the usual quality check on this dataset (slightly different mistakes).
- Create a new variable column called
total_spthat combines all the species. - Select only the
siteandtotal_spvariable into a new dataframe. - Calculate the mean, minimum, maximum, and count of
total_spby site.
Question:
- What is the mean total abundance in site B?
- What is the problem with using mean here? Is there a better way to calculate central tendancy for dount data? (hint: think of decimals in this context).
- Which site has the highest average species abundance?
- Which site has the most replicate samples?
Show answer
str(fake_data)
# there is an inconsistent coding in one of the categorical variables, and an extreme value in one of the numeric variables to remove.
fake_data_clean <- fake_data %>%
dplyr::mutate(site = recode(site, "b" = "B"),
site = as.factor(site)) %>%
dplyr::filter(species_4 < 30) %>%
dplyr::mutate(total_sp = species_1 + species_2 + species_3 + species_4) %>%
dplyr::select(site, total_sp)
fake_data_clean %>%
dplyr::group_by(site) %>%
dplyr::summarise(mean = mean(total_sp),
min = min(total_sp),
max = max(total_sp),
n = n())
# 1. 361.25
# 2. Mode more suitable for integers
# 3. Site B
# 4. site APart 2 - Tidy code
Good coding style is like correct punctuation: you can manage without it, butitsuremakesthingseasiertoread. I mostly comfortable with the tidyeverse syntax, but the most important take away here is keep everything consistent if you prefer a different style.
The full breakdown can be found here.
File names should be machine readable: avoid spaces, symbols, and special characters. Prefer file names that are all lower case, and never have names that differ only in their capitalisation. Delimit words with - or _. Use .R as the extension of R files.
# Good
fit_models.R
utility_functions.R
exploratory-data-analysis.R
# Bad
fit models.R
foo.r
ExploratoryDataAnaylsis.rFile names should be human readable: use file names to describe what’s in the file.
# Good
report-draft-notes.txt
# Bad
temp.rUse the same structure for closely related files:
# Good
fig-eda.png
fig-model-3.png
# Bad
figure eda.PNG
fig model three.pngUse commented lines of - to break up your file into easily readable chunks. Here is a template you can use for your future projects.
# Title.
# Short description of what is being done.
# Authors name and date of analysis.
## Set up ## ----------------------------------------------------------------
# any code related to setting up directory, installing and loading packages (e.g. from workshop 1)
## Loading and cleaning data ## ---------------------------------------------
# any code relating to loading the data, checking the quality, transforming the dataset, so that it is ready for analysis
## Analysis ## -------------------------------------------------------------
# All code involved with preliminary analysis, full analysis, and model output
## Visualisation ## --------------------------------------------------------
# Creating beautiful figures for your report!Object names
Variable and function names should use only lowercase letters, numbers, and underscore. Use underscores (so called snake case) to separate words within a name.
# Good
day_one
day_1
# Bad
DayOne
dayoneGenerally, variable names should be nouns and function names should be verbs. Strive for names that are concise and meaningful (this is not easy!).
# Good
day_one
# Bad
first_day_of_the_month
djm1Where possible, avoid re-using names of common functions and variables. This will cause confusion for the readers of your code.
# Bad
T <- FALSE
c <- 10
mean <- function(x) sum(x)Infix operators
Most infix operators (==, +, -, <-, etc.) should always be surrounded by spaces:
# Good
height <- (feet * 12) + inches
mean(x, na.rm = TRUE)
# Bad
height<-feet*12+inches
mean(x, na.rm=TRUE)There are a few exceptions, which should never be surrounded by spaces:
The operators with high precedence: ::, :::, $, @, [, [[, ^, unary -, unary +, and :.
# Good
sqrt(x^2 + y^2)
df$z
x <- 1:10
# Bad
sqrt(x ^ 2 + y ^ 2)
df $ z
x <- 1 : 10Assignment
Use <-, not =, for assignment.
# Good
x <- 5
# Bad
x = 5Use %>% or |> to emphasise a sequence of actions, rather than the object that the actions are being performed on.
The tidyverse has been designed to work particularly well with the pipe, but you can use it with any code, particularly in conjunction with the _ placeholder.
strings %>%
str_replace("a", "b") %>%
str_replace("x", "y")
strings |>
gsub("a", "b", x = _) |>
gsub("x", "y", x = _)Avoid using the pipe when:
- You need to manipulate more than one object at a time. Reserve pipes for a sequence of steps applied to one primary object.
- There are meaningful intermediate objects that could be given informative names.
Long lines
If the arguments to a function don’t all fit on one line, put each argument on its own line and indent:
# Good
iris %>%
summarise(
Sepal.Length = mean(Sepal.Length),
Sepal.Width = mean(Sepal.Width),
.by = Species
)
iris |>
summarise(
Sepal.Length = mean(Sepal.Length),
Sepal.Width = mean(Sepal.Width),
.by = Species
)
# Bad
iris %>%
summarise(Sepal.Length = mean(Sepal.Length), Sepal.Width = mean(Sepal.Width), .by = Species)For data analysis, we recommend using the pipe whenever a function needs to span multiple lines, even if it’s only a single step.
# Bad
summarise(
iris,
Sepal.Length = mean(Sepal.Length),
Sepal.Width = mean(Sepal.Width),
.by = Species
)Take careful note of the conflicts message that’s printed when you load the tidyverse. It tells you that dplyr overwrites some functions in base R. If you want to use the base version of these functions after loading dplyr, you’ll need to use their full names: stats::filter() and dplyr::filter().
Exercise (15 min)
🧪 Clean the code below
Your colleague got a new job and you have been promoted to his position where you are taking over his unfinished projects. The ecological data has been collected and some quick analysis was performed. But, you noticed that he didnt perform a quality check on the raw data and the # explanations are not clear. The current state is not suitable for the client so you are assigned to clean.
Clean the code where relevent, and check if all the code is working on your end.
Note: change the working directory to your folder and load the plant_calcium.csv to work on.
# Exercise 2
# Cleaning code and tidying dataset
# John Smith, 25-03/2025
## Set up ## ----------------------------------------------------------------
library(tiderVerse)
# Set my directory
setwd("YOUR DIRECTORY")
## Loading and cleaning data ## ---------------------------------------------
# Load raw data
plant data <- read_csv("data/plant_calcium.csv")
str(plant data)
## Analysis ## -------------------------------------------------------------
# Fit model: height explained by elevation
model <- lm(stem_dry_mass ~ stem_length, data = plant_data)
# Check model summary
summary(model)
## Visualisation ## --------------------------------------------------------
# Creating scatterplot figure
plant_data %>%
ggplot() +
geom_point(aes(x = stem_length, y = Stem_dry_mass)) +
labs(
x = "Stem Length (mm)",
y = "Stem Dry Mass (g)",
title = "Stem Dry Mass vs. Stem Length in Sugar Maple Seedlings",
subtitle = "Hubbard Brook LTER"
) +
theme_minimal()Show answer
# Exercise 2
# Cleaning code and tidying dataset
# John Smith, 25-03-2025
## Set up ## ----------------------------------------------------------------
library(tidyverse)
# Set my directory
setwd("YOUR DIRECTORY")
## Loading and cleaning data ## ---------------------------------------------
# Load raw data
plant_data <- read_csv("data/plant_calcium.csv")
str(plant_data)
# Correct class type
plant_data <- plant_data %>%
mutate(watershed = as.factor(watershed),
elevation = as.factor(elevation))
# note, 'transect' does not need to be a factor as there are no distinct categories for transect. It is just text to keep track of replications.
# Check for missing values in each column
colSums(is.na(plant_data))
# Visualise missing data pattern
visdat::vis_dat(plant_data)
# Remove NA rows based on missing elevation data
plant_data <- plant_data %>%
drop_na(elevation)
# Check for duplicate rows
sum(duplicated(plant_data)) # None
# Check for outliers
ggplot(plant_data, aes(x = leaf2area)) +
geom_histogram() +
theme_bw()
# create a clean dataset after filtering the outlier
plant_data_clean <- plant_data %>%
dplyr::filter(leaf2area < 90)
## Analysis ## -------------------------------------------------------------
# Fit model: height explained by elevation
dry_length_mod <- lm(stem_dry_mass ~ stem_length, data = plant_data_clean)
# Check model summary
summary(dry_length_mod)
## Visualisation ## --------------------------------------------------------
# Creating scatterplot figure
plant_data_clean %>%
ggplot() +
geom_point(aes(x = stem_length, y = stem_dry_mass)) +
labs(
x = "Stem Length (mm)",
y = "Stem Dry Mass (g)",
title = "Stem Dry Mass vs. Stem Length in Sugar Maple Seedlings",
subtitle = "Hubbard Brook LTER"
) +
theme_minimal()Assessment
Task to complete before the end of the workshop.
Fortunately, the authors of the study cleaned the data so we do not need to do much data transformation before analysis. For the purpose of future workshops, please select only the following columns: year, watershed, elevation, stem_length, leaf_dry_mass, and stem_dry_mass.
Next, calculate the total dry mass from leaf_dry_mass and stem_dry_mass and correct the unit from kilogram to gram.
Create a separate object to that summarises the mean, standard deviation, and count plant_task_data_clean of the total dry mass variable by watershed and year.
Save this workflow for future workshops. Remember to create a header with the next workshop titled “Week 4 workshop - Descriptive statistics”, a description called “Run some basis statistical summarise and plotting the data”, your name, and the next workshops date.
For the adventurous folks
You’ve learned how to use the core dplyr verbs — now let’s practice building more powerful and efficient workflows by combining multiple functions in clean, compact code.
1. Take your cleaned dataset plant_task_data_clean and write one pipeline that:
- Filters only
W1watershed and year2004. - Creates a new variable
total_mass_gfromstem_dry_mass+leaf_dry_mass, multiplied by 1000 - Selects only the new variable and
stem_length - Filters any rows with
stem_lengthgreater than 200 mm. - Arranges the result by
total_mass_gin descending order.
Use %>% or |> and avoid creating intermediate objects.
2. Using group_by() and summarise() in a single chain:
- Group the data by
watershedandyear. - Calculate the mean, coefficient of variation, and count of
total_mass_g. - Arrange by year in ascending order.
3. Write a function that:
- Takes a numeric column as input.
- Filters out outliers using the 1.5 × IQR rule.
- Returns the filtered data.
Try applying it to leaf_dry_mass and stem_length.
Extra Stuff
- More comprehensive description of the data transformation functions to explore.
- An animated version that visualises what some of the tidyverse functions do.
inner_join() combines two dataframes by a common ‘key’. This function returns only rows with matching keys in both dataframes. Rows without a match are excluded.
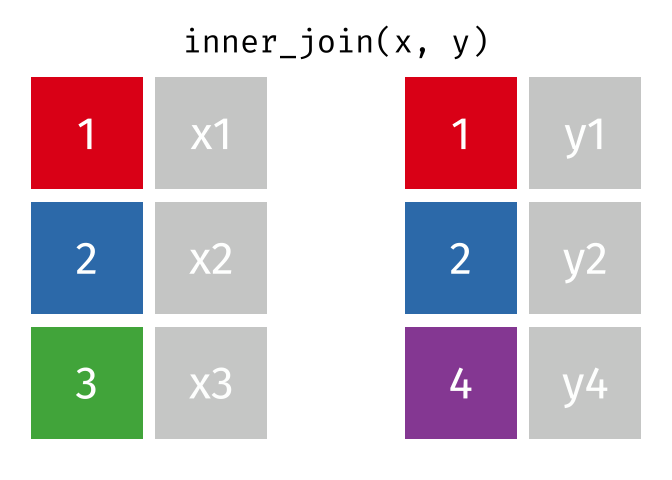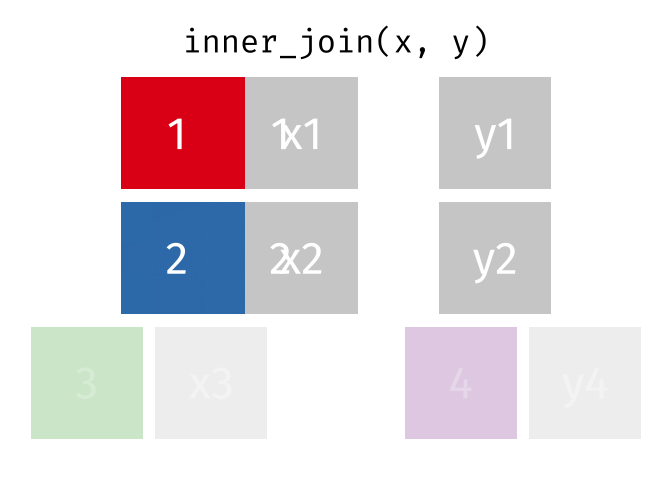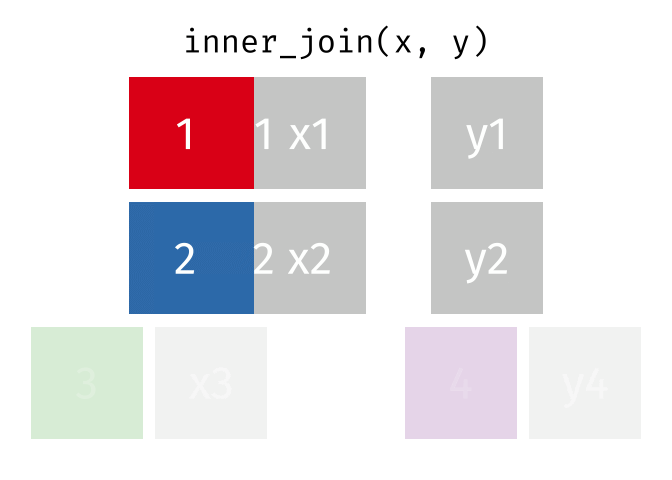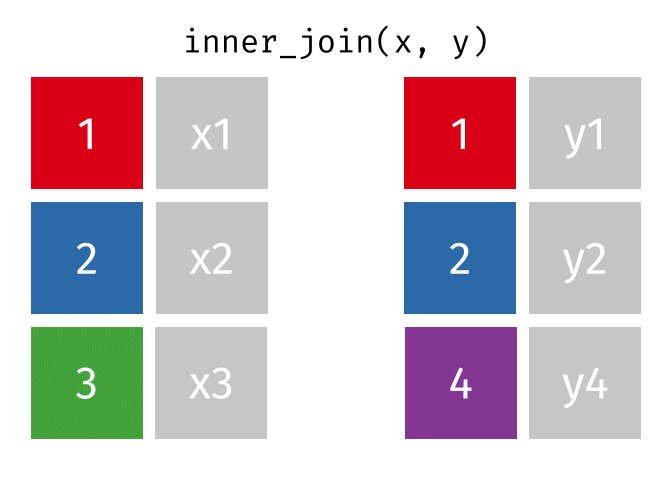
# create two dataframes
df1 <- data.frame(ID = c(1, 2, 3), Name = c('A', 'B', 'C'))
df2 <- data.frame(ID = c(2, 3, 4), Score = c(80, 90, 70))
# apply rbind() function
dplyr::inner_join(df1, df2, by = 'ID')left_join() combines two dataframes by ‘key’. This function returns all rows from the left dataframe, and adds matching rows from the right (unmatched = NA) Rows without a match are excluded.
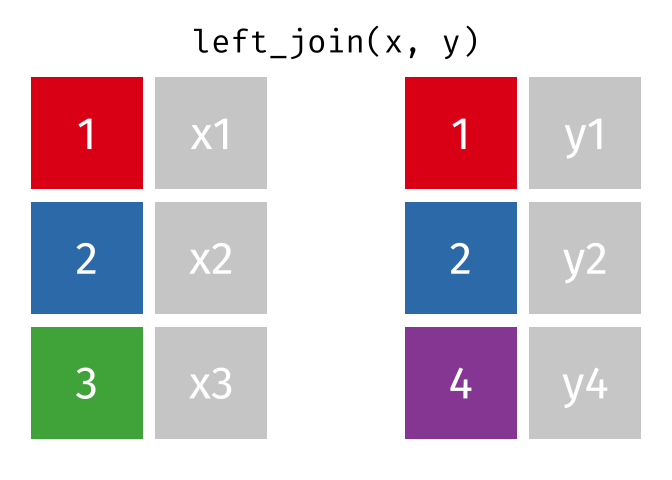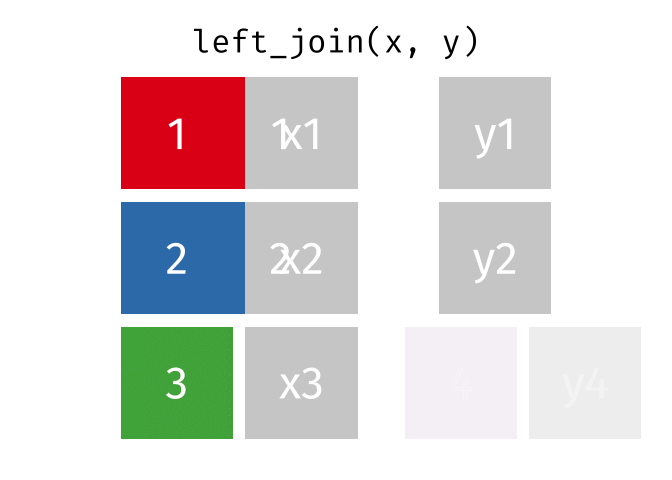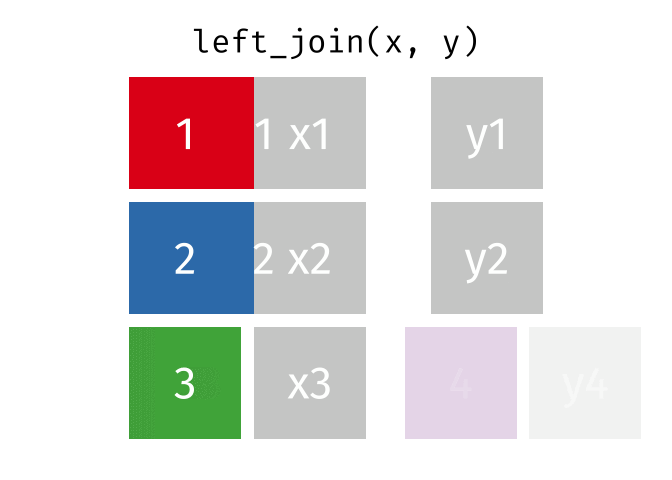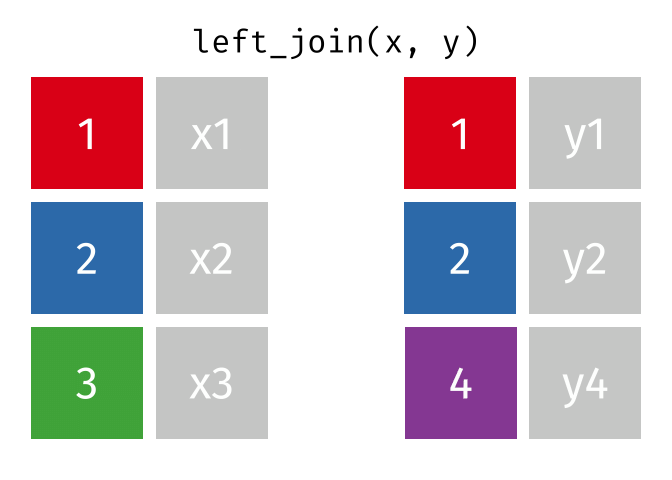
# create two dataframes
df1 <- data.frame(ID = c(1, 2, 3), Name = c('A', 'B', 'C'))
df2 <- data.frame(ID = c(2, 3, 4), Score = c(80, 90, 70))
# apply rbind() function
dplyr::left_join(df1, df2, by = 'ID')right_join() combines two dataframes by ‘key’. This function returns all rows from the right dataframe, and adds matching rows from the left (unmatched = NA) Rows without a match are excluded.

# create two dataframes
df1 <- data.frame(ID = c(1, 2, 3), Name = c('A', 'B', 'C'))
df2 <- data.frame(ID = c(2, 3, 4), Score = c(80, 90, 70))
# apply rbind() function
dplyr::right_join(df1, df2, by = 'ID')full_join() combines two dataframes by ‘key’. This function returns all rows from the both dataframes. The unmatched rows get NA values for missing columns.

# create two dataframes
df1 <- data.frame(ID = c(1, 2, 3), Name = c('A', 'B', 'C'))
df2 <- data.frame(ID = c(2, 3, 4), Score = c(80, 90, 70))
# apply rbind() function
dplyr::full_join(df1, df2, by = 'ID')- Basic data manipulation.
- Efficient data manipulation (streamline your code).
- Advanced data manipulation (diverse
dplyrfunctions).